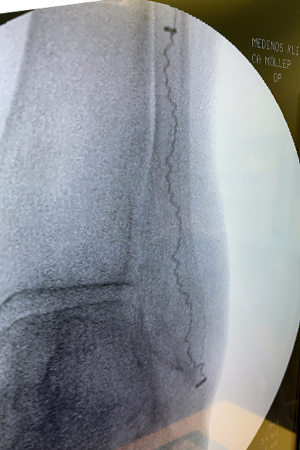
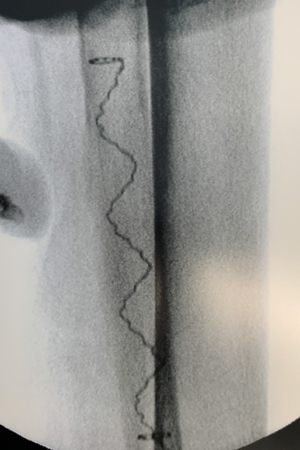
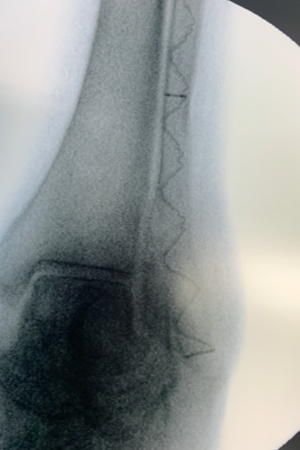
IlluminOss was used to treat a 97-year-old patient with a Weber B fracture of the left fibula after they suffered a fall. A small incision was made at the distal lateral malleolus and a pathway with a 4.0mm awl was created to allow the canal to be prepared with flexible burrs delivered over a 1.2mm guidewire of 5.5mm and 6.5mm diameter to a depth of approximately 50mm beyond the entry pathway.
A 9mm x 120mm implant, with a 4.5mm sheath was delivered within the canal and infused with monomer expanding the implant and filling the canal. The implant was then cured with visible blue light from the IlluminOss lightbox stabilizing the fracture. The incision was closed with two stitches. To reduce any patient discomfort upon their waking up from anesthesia this surgeon injects a small amount of Novocain as a local anesthetic around and under the sutures. At day one post op the patient was walking and was discharged later in the day.