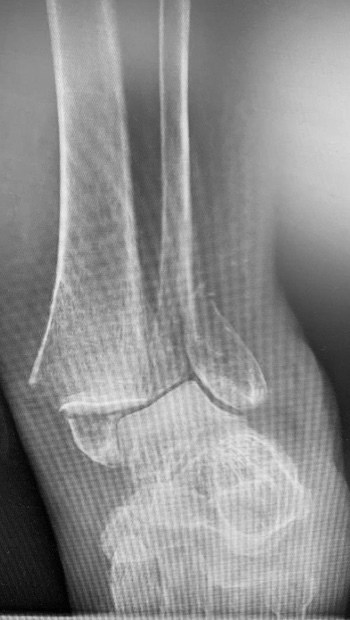
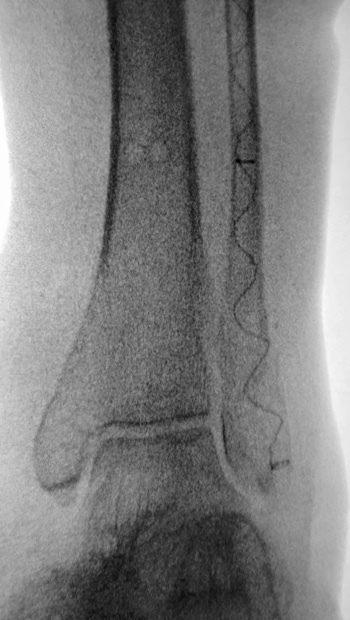
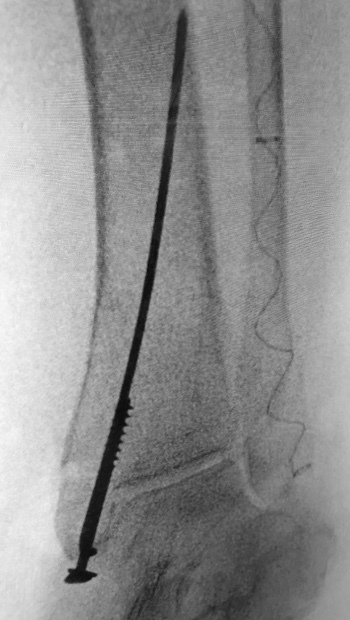
77-year-old female who slipped, fell, and suffered a bi-malleolar fracture. The patient had multiple co-morbidities including osteoporosis. The fibula was stabilized percutaneously using an IlluminOss 9 x 120mm implant delivered through a 4.5mm entry point. The medial malleolus was fixed with a cannulated screw, washer, and K-wire. Post op day 1, the patient was allowed to bear weight as tolerated in an inflatable walker boot.
Download PDF | 900625-B